This page contains the detailed and easy notes for GCSE CCEA Chemistry Organic Chemistry for revision and understanding Organic Chemistry.
Banner 1
GCSE CCEA Chemistry Organic Chemistry Complete Revision Summary
ORGANIC CHEMISTRY
Organic Chemistry
- Hydrocarbons and Crude Oil
- Alkanes
- Fractional Distillation
- Properties of Hydrocarbons
- Cracking
- Alkenes
- Reaction of Alkenes
- Alcohols
- Carboxylic Acid
- Addition Polymerization
- Condensation Polymerization
- Amino Acids
- DNA
Banner 2
CRUDE OIL
- It is a black thick liquid which takes millions of years to form.
- It is the mixture of hydrocarbon.
- Hydrocarbon are the compounds made up of carbon and hydrogen only.
- The components of the crude oil are important and the crude oil is separated by the process of fractional distillation.
HYDROCARBON PROPERTIES

Banner 3
FRACTIONAL DISTILLATION OF CRUDE OIL
- Separating the mixtures on the basis of boiling points.
- It is separated in fractionating column with different substances of similar boiling points
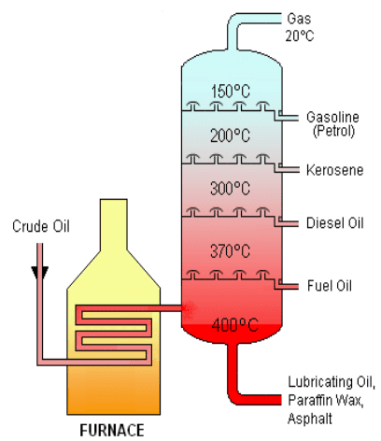
| LIQUIFIED GAS | FUEL |
| GASOLINE/PETROL | CAR FUEL |
| KEROSENE | AIRCRAFT FUEL |
| DIESEL OIL | FUEL IN DIESEL ENGINES |
| RESIDUE | MAKING ROADS |
L – Look
G – Great
K – Kid
D – Doing
R – Roll
CRACKING –Thermal decomposition of longer chain hydrocarbon into a shorter chain alkane and alkenes
Thermal Cracking Catalytic Cracking
It is done at a very high temperature It is done using a catalyst

Banner 3
Organic Chemistry (1)
GCSE CCEA Chemistry ORGANIC CHEMISTRY Complete Revision Summary
FREE DOWNLOAD
Send download link to:
GCSE CCEA Chemistry ORGANIC CHEMISTRY Complete Revision Summary
Send download link to:
WHY CRACKING
- Shorter chain alkanes are more in demand as they are more efficient fuel which fractional distillation alone cannot meet.
- Alkenes are required for polymerization and synthesize other hydrocarbons which fractional distillation cannot meet.
ALKANES – Saturated Hydrocarbon
Carbon-carbon single bond made up of carbon and hydrogen
General Formulae CnH2n+2
Methane – CH4
Ethane – C2H6
Propane – C3H8
Butane – C4H10
Pentane – C5H12
Homologous Series – Members of the same family have similar functional group similar chemical properties and general formulae but different physical property and each members differs from successive by CH2.
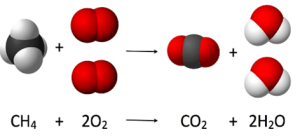
COMBUSTION
COMPLETE
| INCOMPLETE
|
| FUEL IS COMPLETELY BURNED | FUEL IS PARTIALLY BURNED DUE TO LIMITED SUPPLY OF OXYGEN
|
| PRODUCES CARBON DIOXIDE AND WATER | PRODUCES CARBON MONOXIDE AND WATER
|
| IT IS NOT TOXIC | CARBON MONOXIDE IS TOXIC AS IT DECREASES. THE OXYGEN CARRYING CAPACITY OF RED BLOOD CELLS
|
PRODUCTS OF COMBUSTION
Carbon Dioxide Test

Limewater Test Carbon Dioxide will turn limewater milky
Water Test



Banner 4
FUNCTIONAL GROUPS
Groups of atoms that give special properties and reactions to the organic molecule
| Functional Groups | Examples | Formation | |
| ALKENES | = | Ethene, propene, butene, pentene | Cracking of crude oil |
| ALCOHOLS | -OH | methanol, ethanol, propanol, butanol, pentanol | Reaction of alkene with water |
| CARBOXYLIC ACID |  | methanoic acid, ethanoic acid, propanoic acid, butanoic acid. | Oxidation of alcohols |
| ESTERS | 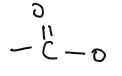 | methyl ethanoate, ethyl ethanoate | Reaction of alcohols and carboxylic acid |
ALKENES
Unsaturated Hydrocarbon
- Compounds which have carbon-carbon double bond
- Compounds made up of carbon and hydrogen only
GENERAL FORMAULE CnH2n
Useful to make polymers, alkanes, alcohols

MANUFACTURE OF ETHANOL
| FERMENTATION | HYDRATION OF ETHENE | |
| REACTION | Glucose Ethanol + carbon dioxide C6H12O6 2C2H5OH + 2CO2 | Ethene + Steam Ethanol |
| REACTION CONDITIONS | Gentle temperature and pressure. Anaerobic conditions | Nickel catalyst and high temperature and pressure |
| ADVANTAGES | Uses renewable resources like sugarcane. Less dependent on fossil fuels and due to less energy requirements do not harm the environment. | It is a continuous process. It is rapid more efficient and have 100% atom economy. Produces more pure ethanol |
| DISADVANTAGES | It is a batch process. The ethanol has to be distilled from time to time as high concentration will kill the yeast. The reaction is slow and produces impure ethanol. Also the atom economy is not 100% | Requires ethene which is dependent on crude oil. Uses non renewable resources. |
Banner 5
REACTIONS OF ALKENES

ALCOHOLS – Have functional Group –OH
General Formulae
CnH2n+1 OH
Formed by replacing hydrogen of alkane with OH group
Used as fuel, solvents, spirits


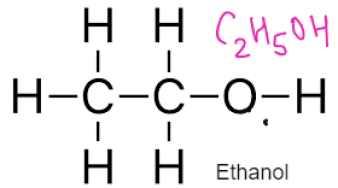

REACTIONS OF ALCOHOLS
COMBUSTION
- It can undergo complete or incomplete combustion. Complete combustion produces carbon dioxide and water.
- Ethanol + Oxygen = Carbon dioxide + water
C2H5OH + O2 CO2 + H2O
- Incomplete combustion produces carbon dioxide and water.
- Ethanol + Oxygen = Carbon monoxide + water
C2H5OH + O2 CO + H2O
OXIDATION
- Alcohols are oxidised to carboxylic acid in the presence of oxidising agent.
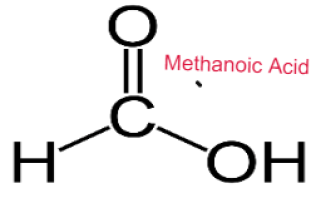
- Methanol Methanoic Acid
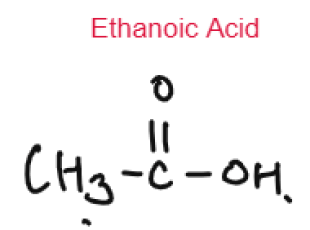
- Ethanol Ethanoic Acid
- Oxidising agent used is acidified potassium dichromate solution
METAL
Alcohols react with metals to form salt and hydrogen.
2C2H5OH + 2Ca 2C2H5OCa + H2
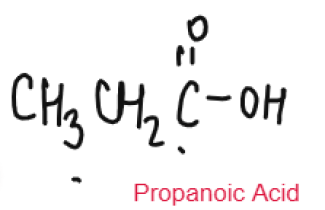
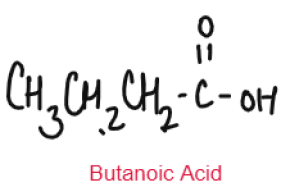
CARBOXYLIC ACID
Weak Acids
Carboxylic Acids are weak acids as they are partially dissociated in water to release H+ ions.
CH3COOH ⇌ CH3COO– + H+
Metal oxides and Metal hydroxide
Carboxylic Acid reacts with metal oxides and metal hydroxide to form salt and water.
CH3COOH + NaOH CH3COONa + H2O
Metal carbonate
Carboxylic Acid reacts with metal carbonate to form salt, water and carbon dioxide.
CH3COOH + Li2CO3 CH3COOLi + CO2 + H2O
Baneer 6
ESTERS
- Fruity smelling compounds
- Used in the manufacture of perfumes, foods and cosmetics.
CARBOXYLIC ACID + ALCOHOLS ESTERS + WATER
Alkyl alkanoate
Methanoic Acid + Methanol Methyl methanoate + Water

ADDITION POLYMERIZATION
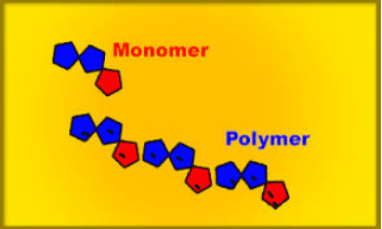

- The individuals unit that polymerizes to form a polymers is known as a monomers. Eg Ethene
- The structure formed by the polymerization of the monomer is a polymers.
Polymers are materials made by linking up smaller repeating chemical units.
Some bend and stretch – rubber and polyester.
Some hard and tough – epoxies and glass.
ADDITION POLYMERS
- a) Formed by addition reaction.
- b) Require only one monomer generally an alkene
- c) Nothing is lost in the reaction.
eg Polyethene, polypropene
CONDENSATION POLYMERS
- a) Requires two monomers
- b) Requires two functional group
- c) Formed by condensation reaction.
- d) A small molecule of water is
- e) Example: Nylon a polyester
NATURAL POLYMERS
- a) They are found naturally
- b) All the complex biomolecules are polymers
| Monomer | Polymer |
| Glucose | Starch |
| Proteins | Amino Acid |
| Nucleotide | DNA |

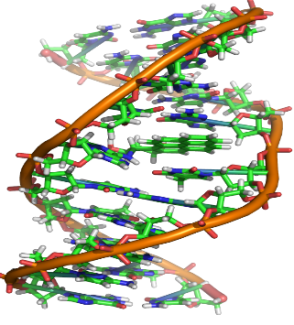
DNA
- a) DNA is polynucleotide
- b) Nucleotide = Phosphate + Sugar + Nitrogenous Bases
- c) There are four bases present in the DNA
- Adenine
- Thymine
- Guanine
- Cytosine

KEY TERMS
Hydrocarbon – Hydrocarbon are the compounds made up of carbon and hydrogen only.
Crude Oil – It is a black thick liquid which takes millions of years to form. It is the mixture of hydrocarbon.
Fractional Distillation – Separating the mixtures on the basis of boiling points.
Alkanes – Saturated Hydrocarbon. Carbon-carbon single bond. Made up of carbon and hydrogen only
Saturated hydrocarbon – Saturated Hydrocarbons have only carbon-carbon single bonds.
Unsaturated hydrocarbon – Unsaturated Hydrocarbons have carbon-carbon double bonds and triple bonds.
General Formula – It applies to families of compounds; provides a way to predict the molecular formula of the molecule, based on the number of carbon atoms it contains.
Viscosity – movement of flow. A fluid with low viscosity flows easily
Flammable – Flammable materials are combustible materials that can easily ignite at room temperature
Complete Combustion – Fuel is completely burned. Produces Carbon dioxide and water.
Incomplete Combustion – Fuel is partially burned due to limited supply of oxygen. Produces Carbon Monoxide and Water.
Cracking – Thermal decomposition of longer chain hydrocarbon into a shorter chain alkane and alkenes
Alkenes – Unsaturated Hydrocarbon. Compounds which have carbon-carbon double bond. Compounds made up of carbon and hydrogen only
Functional Group – Groups of atoms that give special properties and reactions to the organic molecule
Homologous Series – Members of the same family have similar functional group similar chemical properties and general formulae but different physical property and each members differs from successive by CH2
Alcohols – Have functional Group –OH. The General Formulae of Alcohols is CnH2n+1 OH. Used as fuel, solvents, spirits
Carboxylic Acid – Carboxylic Acids are weak acids as they are partially dissociated in water to release H+ ions.
Esters – Fruity smelling compounds. Used in the manufacture of perfumes, foods and cosmetics.
Fermentation – Fermentation is a metabolic process that produces chemical changes in organic substrates through the action of enzymes.
Weak Acid – Weak acids are only partially ionized in their solutions.
Monomers – The individuals unit that polymerizes to form a polymers is known as a monomers. Eg Ethene
Polymers – The structure formed by the polymerization of the monomer is a polymers. Polymers are materials made by linking up smaller repeating chemical units.
Some bend and stretch – rubber and polyester.
Some hard and tough – epoxies and glass.
Addition Polymerization – It is the process of repeated addition of monomers with double or triple bonds to form polymers. There is no loss of an atom or a molecule. Ex – PVC, polyethene, Teflon.
Condensation Polymerization – It is a process that involves repeated condensation reactions between two different monomers. There is a loss of a molecule of water, ammonia etc as a by-product. Ex – Nylon, bakelite, silicon.
Monosaccharide – Simplest carbohydrates (single units). They cannot be hydrolyzed into smaller units. Ex – Glucose, fructose.
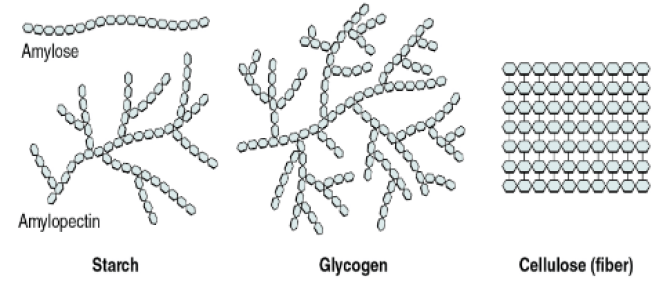
Polysaccharide – Formed of numerous monosaccharide units. Ex – starch, cellulose
Starch – It is the reserve food material of plant cells. It consists of two components- amylose and amylopectin, both glucose polymers.
Cellulose – Main structural polysaccharide of plants. It is a long, unbranched chain of about 6,000 glucose units with molecular weight between 0.5 to 2.5 million.
Proteins – The proteins are linear unbranched polymers of Amino acids. The proteins are composed of carbon, hydrogen, oxygen, nitrogen, and Sulphur.
DNA – It is a long, double chain of deoxyribonucleotide units. DNA is the genetic material and forms molecular basis of heredity in all organisms.
Disclaimer:
I have tried my level best to cover the maximum of your specification. But this is not the alternative to the textbook. You should cover the specification or the textbook thoroughly. This is the quick revision to help you cover the gist of everything. In case you spot any errors then do let us know and we will rectify it.
References:
BBC Bitesize
Wikipedia
Wikimedia Commons
Image Source:
Wikipedia
Wikimedia
Commons
Flickr
Pixabay
Make sure you have watched the above videos and are familiar with the key definations before trying these questions. It is also good to time yourself while doing these questions so that you can work on the speed as well.
Hydrocarbon
- Carbon Compounds as Fuels & Feedstock 1 MS
- Carbon Compounds as Fuels & Feedstock 1 QP
- Carbon Compounds as Fuels & Feedstock 2 MS
- Carbon Compounds as Fuels & Feedstock 2 QP
- Carbon Compounds as Fuels & Feedstock 3 MS
- Carbon Compounds as Fuels & Feedstock 3 QP
Reaction Of Alkenes
- Reactions of Alkenes and Alcohols 1 MS
- Reactions of Alkenes and Alcohols 1 QP
- Reactions of Alkenes and Alcohols 2 MS
- Reactions of Alkenes and Alcohols 2 QP
- Reactions of Alkenes and Alcohols 3 MS
- Reactions of Alkenes and Alcohols 3 QP